What is a Protein? (from PDB-101)
2.92M views716 WordsCopy TextShare
RCSBProteinDataBank
Proteins play countless roles throughout the biological world, from catalyzing chemical reactions to...
Video Transcript:
Proteins play countless roles throughout the biological world. Some transport nutrients throughout the body. Some help the chemical reactions happen at a faster rate.
Others build the structures of the living things. Despite this wide range of functions, all proteins are made out of the same 21 building blocks, called amino acids. Amino acids are made of carbon, oxygen, nitrogen, and hydrogen, and some contain sulfur atoms.
Selenocysteine is the only standard amino acid that contains a selenium atom. These atoms form an amino group, a carboxyl group, and a side chain attached to a central carbon atom. The side chain is the only part that varies from amino acid to amino acid and determines its properties.
Hydrophobic amino acids have carbon-rich side chains, which don’t interact well with water. Hydrophilic or polar amino acids interact well with water. Charged amino acids interact with oppositely charged amino acids or other molecules.
Primary Structure The primary structure of a protein is the linear sequence of amino acids as encoded by DNA. The amino acids in a protein are joined by peptide bonds, which link the amino group of one amino acid to the carboxyl group of another. A water molecule is released each time a bond is formed.
The linked series of carbon, nitrogen, and oxygen atoms make up the protein backbone. These protein chains often fold into two types of secondary structures: alpha helices, or beta sheets. An alpha helix is a right-handed coil stabilized by hydrogen bonds between the amine and carboxyl groups of nearby amino acids.
Beta-sheets are formed when hydrogen bonds stabilize two or more adjacent strands. The tertiary structure of a protein is the three-dimensional shape of the protein chain. This shape is determined by the characteristics of the amino acids making up the chain.
Many proteins form globular shapes with hydrophobic side chains sheltered inside, away from the surrounding water. Membrane-bound proteins have hydrophobic residues clustered together on the outside, so that they can interact with the lipids in the membrane. Charged amino acids allow proteins to interact with molecules that have complementary charges.
The functions of many proteins rely on their three-dimensional shapes. For example, hemoglobin forms a pocket to hold heme, a small molecule with an iron atom in the center that binds oxygen. Two or more polypeptide chains can come together to form one functional molecule with several subunits.
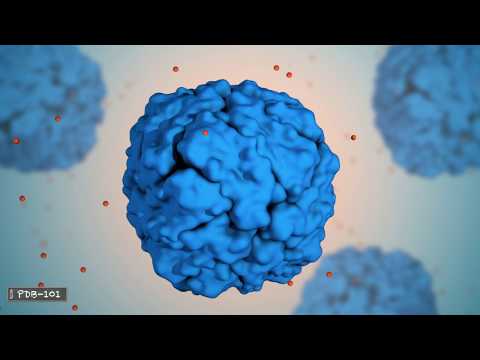
The four subunits of hemoglobin cooperate so that the complex can pick up more oxygen in the lungs and release it in the body. Different visual representations of proteins can give us visual clues about the protein structure and function. This space filling diagram shows all atoms that are making up this protein.
This representation, called ribbon or cartoon diagram shows the organization of the protein backbone and highlights the alpha helices. This surface representation shows the areas that are accessible to water molecules. Most proteins are smaller than the wavelength of light.
For example, the hemoglobin molecule is about 6. 5 nanometers in size. Hemoglobin is found in high concentration in red blood cells.
A typical red blood cell contains about 280 million hemoglobin molecules. The three-dimensional shapes of proteins determine their function. The flexible arms of antibodies protect us from disease by recognizing and binding to pathogens and targeting them for destruction by the immune system.
The hormone insulin is a small, stable protein that can easily maintain its shape while traveling through the blood to regulate the blood glucose level. Alpha Amylase is an enzyme that begins digestion of starches in our saliva. The calcium pump is aided by magnesium and powered by ATP to move calcium ions back to the sarcoplasmic reticulum after each muscle contraction.
Ferritin is a spherical protein with channels that allow the iron atoms to enter and exit depending upon organism’s needs. On the inside ferritin forms a hollow space interior with atoms attached to the inner wall. Ferritin stores iron in the non-toxic form.
Collagen forms a strong triple helix that is used throughout the body for structural support Collagen molecules can form elongated fibrils which aggregate to form collagen fibers. This type of collagen is found in skin and tendons. Learn more about the functions and 3D structures of proteins and other molecular machines at the RCSB Protein Data Bank.
Related Videos
4:52
How Enzymes Work (from PDB-101)
RCSBProteinDataBank
1,422,113 views
3:59
Proteins: Explained
MinuteEarth
223,184 views

6:11
Proteins
Nucleus Biology
96,689 views

19:13
Protein synthesis animation
REDMEDBD
2,426,107 views

5:44
Mitochondria: the cell's powerhouse
Harvard Online
1,066,051 views
2:42
From DNA to protein - 3D
yourgenome
20,642,110 views

6:52
Proteins and their Structure
Aasoka
185,841 views

7:45
Electron Transport Chain
Omar Ali
1,436,474 views

3:39
What is a Protein? Learn about the 3D shap...
RCSBProteinDataBank
973,495 views

7:49
We Are Living in Unprecedented Times
Bernie Sanders
1,079,125 views
4:32
CRISPR: Gene editing and beyond
nature video
1,936,864 views

22:20
How AI Cracked the Protein Folding Code an...
Quanta Magazine
388,058 views
7:46
Protein Structure and Folding
Amoeba Sisters
2,503,082 views
9:09
Animations of unseeable biology | Drew Ber...
TED
2,591,590 views

10:20
The Smartest Way To Use Protein To Build M...
Jeff Nippard
9,259,117 views

5:11
How do carbohydrates impact your health? -...
TED-Ed
6,589,860 views

14:14
How Companies Are Dodging Trump Tariffs On...
CNBC
2,362,624 views

7:28
COVID-19 Animation: What Happens If You Ge...
Nucleus Medical Media
340,284,158 views

12:49
Trudeau: Canada will retaliate with 25% ta...
CBC News
1,315,381 views

7:20
DNA animation (2002-2014) by Drew Berry an...
WEHImovies
6,060,106 views